Actavis Group Hf (Respondent/Claimant) v Eli Lilly & Company
| Jurisdiction | England & Wales |
| Judge | Lord Justice Kitchin,Lord Justice Lloyd,Lord Justice Longmore |
| Judgment Date | 21 May 2013 |
| Neutral Citation | [2013] EWCA Civ 517 |
| Docket Number | Case No: A3/2012/3333 & 3334 |
| Court | Court of Appeal (Civil Division) |
| Date | 21 May 2013 |
[2013] EWCA Civ 517
IN THE COURT OF APPEAL (CIVIL DIVISION)
ON APPEAL FROM THE HIGH COURT OF JUSTICE
CHANCERY DIVISION (PATENTS COURT)
THE HON MR JUSTICE ARNOLD
Royal Courts of Justice
Strand, London, WC2A 2LL
Lord Justice Longmore
Lord Justice Lloyd
and
Lord Justice Kitchin
Case No: A3/2012/3333 & 3334
Mr Stephen Phillips QC and Mr Roger Wyand QC (instructed by Hogan Lovells International LLP) for the Appellant
Mr Richard Meade QC and Mr Thomas Raphael (instructed by Bird & Bird LLP) for the Respondents
Hearing date: 22 April 2013
Approved Judgment
Introduction
This is an appeal from the decision of Arnold J dated 27 November 2012 and his consequential order in a dispute about jurisdiction in two related patent actions. The claimants, Actavis Group hf ("Actavis Group") in the first action, and Medis ehf ("Medis") in the second, are both companies in the Actavis group of companies, a well known supplier of generic pharmaceuticals. I will refer to the claimants collectively as "Actavis".
The defendant, Eli Lilly & Company ("Lilly"), is incorporated in the State of Indiana, USA and is a major research-based pharmaceutical company. It has operating subsidiaries in many countries, including Eli Lilly & Company Ltd ("Lilly UK") which is incorporated in England.
One of Lilly's important products is a cancer treatment called Alimta which contains as its active ingredient a compound called pemetrexed. Alimta was covered by a basic patent which has now reached the end of its term but the protection it afforded to Alimta has been extended by Supplementary Protection Certificates (SPCs) which will expire in December 2015. Lilly also owns European Patent No 1 313 508 ("the Patent") which has designations in the UK, France, Germany, Italy and Spain and which will not expire until 2021. Its claims are, however, directed to the use of pemetrexed in the form of its disodium salt.
Actavis Group, through its various national subsidiaries, wishes to market its own product in competition with Alimta. It accepts that it cannot do so before December 2015 when the SPCs expire. But it contends that it can do so thereafter because its product contains pemetrexed dipotassium and so cannot fall within the scope of the Patent.
In these proceedings Actavis therefore seek a declaration that a product containing pemetrexed dipotassium does not infringe the UK, French, German, Italian or Spanish designations of the Patent. They do not challenge the validity of the Patent, but only want the issue of infringement determined. They say the issue is a short one, that they need its resolution in good time before the expiry of the SPCs and that it would be far more convenient to have it determined in a single trial in England for all the national designations.
Lilly does not accept that a product containing pemetrexed dipotassium does not infringe the Patent. Moreover, it maintains that the issue of infringement is not a simple one and will involve a consideration of the relevant laws of construction (including the so-called doctrine of equivalents) and contributory infringement applying to each designation. Accordingly, although it accepts that this is the appropriate jurisdiction to determine the issue of infringement in relation to the UK designation of the Patent, it says that the issue would more conveniently be determined in relation to the other designations in each of their respective territories.
Lilly therefore challenged jurisdiction in respect of the foreign designations of the Patent, and contended that there had been no proper service of proceedings upon it. This gave rise to three separate issues: first, whether Lilly's solicitors, Hogan Lovells International LLP, had agreed to accept service; second, whether service was properly effected on Lilly under CPR 6.9 at a place of business within the jurisdiction; and third, whether service was properly effected on Lilly under CPR 63.14 on the basis that the claims relate to the UK designation of the Patent. Lilly also raised a fourth issue, namely whether even if (contrary to its case) there had been proper service under CPR 6.9 or CPR 63.14, the court should decline to hear the claims in respect of the foreign designations on the ground of forum non conveniens.
The judge found in favour of Actavis Group in the first action on the first, second and fourth issues; and he found in favour of Medis in the second action on the second and fourth issues. He therefore dismissed Lilly's applications, holding that the court had, and would exercise, jurisdiction to hear the claims in both actions, including the claims in respect of the foreign designations.
The judge gave Lilly permission to appeal to this court on all the issues on which it lost. Actavis have responded that the judge ought also to have found in their favour in both actions on the third issue.
The following issues therefore arise on the appeal:
i) whether the judge was right to hold that Lilly's solicitors, Hogan Lovells, agreed to accept service of proceedings in the first action;
ii) whether the judge was right to hold that service of proceedings in both actions was validly effected under CPR 6.9;
iii) whether the judge ought also to have held that service of proceedings in both actions was validly effected under CPR 63.14;
iv) in the event service was effected under CPR 6.9 or CPR 63.14, whether the judge ought to have granted a stay on the ground of forum non conveniens.
At the outset of the appeal we decided to hear full argument on issues (i), (ii) and (iii) before turning to issue (iv). Having done so, we reached the conclusion that the judge's decision on issue (i) in the first action must be upheld. We also formed the view that this conclusion was dispositive of the whole appeal, the parties having agreed that in these circumstances the second action would become redundant. We communicated these conclusions to the parties and they agreed that our conclusion on issue (i) disposed of the appeal. We also indicated we would provide our reasons in writing in the usual way, and that I now do. Although not necessary for the disposal of this appeal, the parties also invited us to express our conclusions on issues (ii) and (iii) because, so we were told, there is now a third action on foot in which they may be determinative and accordingly our decision upon them would likely avoid the need for a further appeal to this court in that action. I shall therefore express my conclusion on those issues too.
The background
The judge set out the background facts in his judgment from [2]-[24]. The following matters are of particular relevance to the issues with which I must deal and I have drawn them in large measure and with gratitude from the judgment.
I must begin by explaining a little more about the Actavis group of companies. Actavis Group is the parent operating company of the whole group and is incorporated in Iceland. It has a considerable number of subsidiaries incorporated in different countries which are responsible for selling its products. These subsidiaries include Actavis France, Actavis Deutschland GmbH & Co KG, Actavis Italy SpA, Actavis Spain SA and Actavis UK Ltd. Actavis Group PTC ehf ("Actavis PTC") is another subsidiary of Actavis Group and itself has a number of subsidiaries, including Actavis hf and Medis, both of which are incorporated in Iceland. But, as Lilly accepted at the hearing, Actavis PTC does not have relevant subsidiaries in each of France, Germany, Italy and Spain. The relationship between these various Actavis companies is shown below:
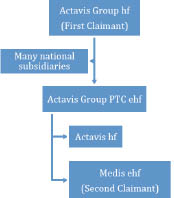
Lilly is the parent company of the Lilly group and is the proprietor of the vast majority of European patents held by the group, including the Patent. Dr Ivan Burnside is identified on the Patent as Lilly's representative for proceedings under the EPC and his address is given as Lilly UK, European Patent Operations, Lilly Research Centre, Erl Wood Manor, Windlesham in Surrey. Lilly has also furnished this address to the Intellectual Property Office as the address for service of proceedings in respect of the UK designation of the Patent under rule 103 of the Patent Rules 2007.
On 12 July 2012, the solicitors for the claimants, Bird & Bird LLP, wrote to Dr Burnside in his capacity as Lilly's representative in the following terms:
" EP 1 313 508 ("the Patent")
We represent Actavis Group PTC ehf and its relevant national subsidiaries ("Actavis").
You are registered as the proprietor of European Patent (UK) No. 1 313 508 B1 entitled "Combination containing an antifolate and methylmalonic acid lowering agent" that is currently valid until 15 June 2021.
The purpose of this letter is to put you on notice that Actavis wishes at the expiry of SPC/GB05/011 to launch a pemetrexed product for use in the manufacture of a medicament for use in combination therapy for inhibiting tumour growth in mammals in the United Kingdom. At the same time (i.e. upon expiry of the equivalent SPCs) Actavis similarly wishes to launch such a product in other jurisdictions, including but not limited to Germany, France, Italy and Spain.
Actavis is aware of the Patent. We should be grateful if you would treat this letter as relating to the national designations of EP 1 313 508 B1 in Germany (DE60127970 (T2)), France (EP 1 313 508 B1), Italy (EP 1 313 508 B1), Spain (ES 2284660 (T3)) and the...
To continue reading
Request your trial-
Fujifilm Kyowa Kirin Biologics Company Ltd v Abbvie Biotechnology Ltd and Another
...which. But FKB's claim cannot be characterised as purely concerned with infringement of the patents (contrast the claim in Actavis Group hf v Eli Lilly & Co [2012] EWHC 3316 (Pat) affd [2013] EWCA Civ 517, [2013] RPC 79 I would add that this analysis makes sense if one considers the possi......
-
Actavis UK Ltd and Others v Eli Lilly & Company
...Spanish designations which I rejected for the reasons given in my judgment dated 27 November 2012, [2012] EWHC 3316 (Pat) (affirmed [2013] EWCA Civ 517, [2013] RPC 37). Since then, there have been a number of developments, of which the following are the most significant for present purpos......
-
Actavis UK Ltd and Others v Eli Lilly & Company
...to hear and determine Actavis' action for DNIs in respect of the foreign designations and his judgment was upheld in this court: [2013] EWCA Civ 517; [2013] RPC 37. This court considered that there was jurisdiction on the ground that Lilly had accepted service of the proceedings. It did no......
-
Actavis UK Ltd and Others v Eli Lilly & Company
...of the Patent (see my judgment dated 27 November 2012 [2012] EWHC 3316 (Pat) and the judgment of the Court of Appeal dated 21 May 2013 [2013] EWCA Civ 517, [2013] RPC 37, " Actavis I"), Lilly counterclaimed for threatened infringement of the UK designation. As explained in more detail belo......
-
The Basic Of Patent Law - Revocation, Non-Infringement And Clearing The Way
...power. It includes, for example, power to make declarations in respect of non-UK designations of European patents (Actavis v Lilly [2013] EWCA Civ 517). Other declaratory relief In Fujifilm Kyowa Kirin Biologics ("FKB") v AbbVie [2017] EWHC 395 (Pat), the Patents Court awarded, in a landmar......
-
Technical Matters And Procedure - Annual Patents Review 2017
...& Ors [2017] UKSC 48 Actavis Group HF v Eli Lilly and Company [2012] EWHC 3316 (Pat); Actavis Group hf v Eli Lilly & Company [2013] EWCA Civ 517 Parainen Pearl Shipping Limited & Ors v Kristian Gerhard Jebsen Skipsrederi AS & Ors [2017] EWHC 2570 (Pat) (18 October 2017) Arno......
-
English Court Will Exercise Jurisdiction Over Foreign Designations Of European Patents Where Validity Is Not Challenged
...Group and Eli Lilly & Company [2013] EWCA Civ 517 The Court of Appeal has recently dismissed a jurisdictional challenge brought by Eli Lilly in respect of the non-UK designations of a European Patent. In reaching their decision, the Court of Appeal has confirmed that the English courts ......
-
Court Of Appeal Confirms Cross-Border Non-Infringement Jurisdiction
...or regional division of the UPC. 1) In joined cases Actavis Group HF v Eli Lilly and Company and Medis EHF v Eli Lilly and Company [2013] EWCA Civ 517. The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your spe......
-
Cross-border litigation in England and Wales
...EWCA Civ. 555.76. (England and Wales) Actavis Group v. Eli Lilly & Co, [2012] EWHC 3316 affirmed in Actavis Group hf v. Eli Lilly &Co, [2013] EWCA Civ. 517.152 Maastricht Journal of European and Comparative Law the desired legal remedies to the claiman ts. Although the Court of Appeal77set ......

