Medimmune Ltd v Novartis Pharmaceuticals UK Ltd
| Jurisdiction | England & Wales |
| Judge | Lord Justice Kitchin,Lord Justice Lewison,Lord Justice Moore-Bick |
| Judgment Date | 10 October 2012 |
| Neutral Citation | [2012] EWCA Civ 1234 |
| Docket Number | Case Nos: A3/2011/2562/ A3/2012/0626 |
| Court | Court of Appeal (Civil Division) |
| Date | 10 October 2012 |
[2012] EWCA Civ 1234
Lord Justice Moore-Bick
Lord Justice Lewison
and
Lord Justice Kitchin
Case Nos: A3/2011/2562/
A3/2011/2562(Y) and
A3/2012/0626
IN THE COURT OF APPEAL (CIVIL DIVISION)
ON APPEAL FROM THE HIGH COURT OF JUSTICE
CHANCERY DIVISION (PATENTS COURT)
The Hon Mr Justice Arnold
HC09C04770/HC11C01304
Royal Courts of Justice
Strand, London, WC2A 2LL
Roger Wyand QC, Richard Meade QC and Tom Mitcheson (instructed by Marks & Clerk Solicitors LLP) appeared on behalf of MedImmune Limited
Simon Thorley QC, Justin Turner QC and Joe Delaney (instructed by Allen & Overy LLP) appeared on behalf of Novartis Pharmaceuticals UK Limited
Hearing dates : 9–11 July 2012
Introduction
These appeals concern two judgments of Arnold J given in related proceedings. In action HC09C04770 MedImmune Limited ("MedImmune"), formerly known as Cambridge Antibody Technology Limited ("CAT"), alleged Novartis Pharmaceuticals UK Limited ("Novartis") had infringed European Patents (UK) Nos. 0 774 511 and 2 055 777 ("the 777 patent" or "the patent") by selling a product called ranibizumab which is used for the treatment of wet age-related macular degeneration of the eye. Novartis disputed infringement and counterclaimed for revocation of the patents.
The judge held that both patents were invalid on the grounds of obviousness and because the claims were not entitled to priority from the relevant priority document, it being conceded that if they were not entitled to priority then the patents were invalid. He also held the claims did not cover the process by which ranibizumab was made. MedImmune was granted permission to appeal against the judge's findings in respect of both patents but has only pursued the appeal in respect of the 777 patent. It contends the judge erred in making each of these findings. By way of respondent's notice, Novartis contends the judge ought additionally to have found the 777 patent invalid for insufficiency, and that it was not infringed for the further reason that ranibizumab is not "obtained directly" by means of the claimed process.
In the second action HC11C01304, Novartis sought a declaration that a Supplementary Protection Certificate ("the SPC") granted in respect of the 777 patent was invalid. It necessarily followed from the judge's findings in the first action that the SPC was invalid but the judge held that even on the hypothesis that his earlier findings were wrong, the SPC was nonetheless invalid because it was granted in respect of a product which was not identified in the wording of the relevant claim as a product deriving from the process in question. MedImmune appeals against that further finding.
Upon these appeals we have had the benefit of a scientific advisor, Professor Brian Henderson of University College London. Professor Henderson is Professor of Biochemistry at the UCL-Eastman Dental Institute. Prior to the hearing he gave us three "teach-ins" about the background technology. During the hearing he sat with us and provided us with further assistance in understanding the technical issues. He has also read through this judgment in draft for technical accuracy. At no time has Professor Henderson expressed any opinion to us about any of the matters in dispute. He has been immensely helpful and we are very grateful to him.
At the outset of these appeals we decided to hear full argument on the obviousness and priority issues. Having heard those arguments, we reached the clear conclusion that the judge's decision on obviousness must be upheld and so informed the parties. We also indicated that it appeared to us that conclusion was dispositive of both appeals and that it would not be in the interests of justice to hear further argument on all the other issues. The parties agreed. We also indicated we would provide our reasons in writing in the usual way. That I now do.
The patented technique – an outline
The 777 patent claims priority from a number of applications, but the only one of relevance to these proceedings was filed on 12 November 1990. The patent is concerned with a technique called antibody phage display. The invention described in the patent was developed by CAT (as MedImmune was then known) in close co-operation with scientists at the Medical Research Council ("the MRC") and the patent is therefore jointly owned by MedImmune and the MRC. The MRC was joined to the actions so as to be bound by their result, but has played no part in the proceedings.
Antibody phage display provides a particularly advantageous way of selecting desirable antibody fragments based upon their ability to bind to particular antigens. The method makes use of particular filamentous bacteriophages. Bacteriophages, often referred to simply as phages, are viruses which infect bacteria. They consist of a protein coat or capsid encapsulating nucleic acid. Filamentous phages are non-lytic, meaning they are extruded from an infected bacterial cell which continues to live and produce more phages. Filamentous phages may be distinguished from lytic phages, such as lambda phages, which are only released when the host cell bursts.
A filamentous phage consists of a single-stranded DNA genome packed in a long tube composed of a protein coat. One end comprises two proteins, one of which is called pIII. This particular protein plays a vital role in the process of infection. It has three domains; two N terminal domains extend from the phage surface, and the third, the C terminal domain, is buried in the phage particle. The phage infects a bacterial cell through long slender proteinaceous appendages called pili on the cell surface. The pilus is first bound by the N2 domain of pIII and then the N1 domain of pIII binds to a bacterial surface protein. The phage DNA is then inserted into the bacterium's cytoplasm where it is used to make more DNA and for protein expression. When enough DNA and protein have been generated, production of new phage particles begins. Assembly of the new phages occurs in the cytoplasmic membrane of the bacterium and various non-coat proteins create a channel for the passage of each nascent phage out of the cell. The DNA is extruded into the phage body, the capsid completed and the phage released from the bacterial membrane.
The 777 patent describes the generation of filamentous phage particles that have an antibody fragment displayed on their surface by fusion to one of the viral coat proteins, particularly the pIII protein, while the DNA sequence encoding that antibody fragment is included within the genome inside the phage particle. The displayed antibody fragment is correctly folded and functional when fused to the viral coat protein, so that it can bind to its antigen or, more precisely, its target epitope. The display of the antibody fragment on the phage surface means that the ability of the displayed antibody fragment to bind to an antigen can be tested in vitro. Typically antigen is immobilised on a solid substrate and then presented with a library of potential binders, the phage antibody library. Those antibody fragments that bind to the antigen will attach to the immobilised antigen while those that do not bind can be washed away. Once the non-binders have been washed away, the selected phage antibody can be released from the antigen by a process known as elution. The essential elements of the process are depicted diagrammatically below:
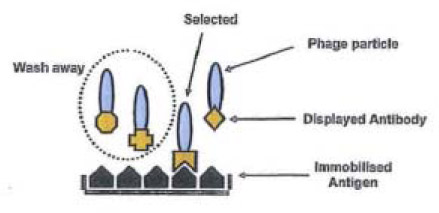
One of the key features of phage display is that each antibody fragment is physically associated with the nucleic acid encoding its sequence which is contained within the phage upon which the antibody fragment is displayed. Having selected the relevant phage antibody, the sequence of the antibody fragment polypeptide can readily be elucidated by sequencing the DNA of the phage which displayed it. Further, the technique allows scientists to screen very large libraries of different antibody fragments.
The technical background
The invention required the bringing together of developments in two technologies, antibody engineering and phage display.
Antibodies are protein molecules which are generated by an animal's immune system to assist in neutralising or destroying foreign matter, for example bacteria and viruses. The surface of bacteria and viruses contains proteins. When such foreign proteins enter the bloodstream, the immune system of the host recognises them as being foreign and sets about trying to destroy or neutralise them by producing antibodies. The individual foreign molecules are known as antigens and the sites in these molecules recognised by the antibodies are called epitopes. Each antibody locks on to a single epitope but, since each epitope is different, different antibodies have to be made to lock on to each of them.
The structure of antibodies, the way they are generated and their functions are described in the technical primer agreed between the parties and at [39] – [62] of the judgment. For the purpose of this appeal, I would emphasise the following.
Antibodies are generated by specialist cells called B lymphocytes. Each cell can produce only a single design of antibody. Nevertheless, humans have the ability to generate a vast array of different antibodies, in part by the rearrangement of multiple small genes...
To continue reading
Request your trial-
Eli Lilly and Company v Janssen Alzheimer Immunotherapy
...prepare a technical primer for the court. In my view a technical primer of the kind which was prepared by the parties in MedImmune Ltd v Novartis Pharmaceuticals UK Ltd [2011] EWHC 1669 (Ch) would have been of assistance. I appreciate that agreeing a technical primer is not always easy, and......
-
Saab Seaeye Ltd v Atlas Elektronik GmbH and Another Adrian Dann and Others (Third Parties)
...the obviousness attacks on both patents. 83 Most of the principles can be extracted from the judgments of Kitchin and Lewison LJJ in Medimmune v Novartis [2012] EWCA Civ 1234. "84. The starting point is that an invention must involve an inventive step, and it is to be taken to involve an in......
-
BDI Holding GmbH v Argent Energy Ltd
...as make a précis.” 20 Use of the inventive concept for assessing obviousness is optional, but Kitchin LJ pointed out in Medimmune Ltd v Novartis Pharmaceuticals UK Ltd [2012] EWCA Civ 1234; [2013] RPC 27 at [86]: “… it has the advantage of limiting the obviousness analysis to the essence ......
-
Samsung Electronics Company Ltd v Apple Retail Uk Ltd and Another
...referring to the input frame and cannot be referring to the superframe. 135 Entitlement to priority 136 The law 137 In Medimmune Limited v Novartis Pharmaceuticals Limited the Court of Appeal summarised the law on entitlement to priority in this way: "151. Section 5(2)(a) of the Patents Act......
-
IP Bulletin - November 2012
...The question of obviousness – observations from the Court of Appeal Medimmune Ltd v Novartis Pharmaceuticals UK Ltd and another [2012] EWCA Civ 1234, 10 October In Medimmune Ltd v Novartis Pharmaceuticals UK Ltd and another [2012] EWCA Civ 1234, 10 October 2012, the Court of Appeal has uphe......
-
Court Of Appeal Confirms Invalidity Of Patent For EXELON Because It Was Obvious To Resolve Racemate Using Standard Techn
...fair expectation of success within a reasonable timeframe (eg Kitchin LJ in Medimmune Ltd v Novartis Pharmaceuticals UK Ltd & Ors [2012] EWCA Civ 1234). The Novartis contended that the High Court had incorrectly used the "obvious to try" test and had not considered whether there was a f......
-
Obviousness - Recent Cases Of The Court Of Appeal
...a reasonable expectation of success" test. Kitchin LJ disagreed, emphasising with reference to his decision in MedImmune v Novartis [2012] EWCA Civ 1234 that the statutory question, "was the invention obvious?", should not be paraphrased. He stated I reject the submission that the court can......
-
Validity - Annual Patents Review 2017
...& Ors [2017] EWCA Civ 266 (12 April 2017) Gross LJ, Floyd LJ, Arnold J MedImmune Limited v Novartis Pharmaceuticals UK Limited [2012] EWCA Civ 1234 Biogen Inc v Medeva Plc [1996] UKHL 18 Teva UK Ltd v Leo Pharma A/S [2015] EWCA Civ 779 Actavis Group PTC EHF & Anr v ICOS Corporation ......

