Gilead Sciences Inc. (a company registered in the US State of Delaware) v Nucana Plc
| Jurisdiction | England & Wales |
| Judge | Mr Justice Meade |
| Judgment Date | 21 March 2023 |
| Neutral Citation | [2023] EWHC 611 (Pat) |
| Court | Chancery Division (Patents Court) |
| Docket Number | Case No: HP-2021-000007 |
[2023] EWHC 611 (Pat)
The Hon. Mr. Justice Meade
Case No: HP-2021-000007
IN THE HIGH COURT OF JUSTICE
BUSINESS AND PROPERTY COURTS OF ENGLAND AND WALES
INTELLECTUAL PROPERTY LIST (ChD)
PATENTS COURT
The Rolls Building
7 Rolls Buildings
Fetter Lane
London EC4A 1NL
Michael Tappin KC, Tom Moody-Stuart KC and James Whyte (instructed by Bird & Bird LLP) for the Claimants/Part 20 Defendants
Piers Acland KC, Tom Mitcheson KC and Andrew Lomas (instructed by Powell Gilbert LLP) for the Defendant/Part 20 Claimant
Hearing dates: 20 and 23–27 January and 1–3 February 2023
APPROVED JUDGMENT
Remote hand-down: This judgment will be handed down remotely by circulation to the parties or their representatives by email and release to The National Archives. A copy of the judgment in final form as handed down should be available on The National Archives website shortly thereafter but can otherwise be obtained on request by email to the Judicial Office (press.enquiries@judiciary.uk).
| Introduction | 5 |
| EPO timing | 8 |
| Relevance of Idenix v Gilead | 9 |
| The witnesses | 10 |
| Professor Micklefield – Gilead's medicinal chemistry expert | 10 |
| Doctor Galmarini – Gilead's oncology expert | 10 |
| Prof Matthias Götte | 11 |
| Professor Seley-Radtke – NuCana's medicinal chemistry expert (non-synthesis) | 11 |
| Professor Smyth – NuCana's oncology expert | 12 |
| Professor Davies – NuCana's synthetic chemistry expert | 12 |
| Prof Davies' instructions for the EPO | 14 |
| The skilled team | 16 |
| CGK | 17 |
| Approach to CGK – the law | 17 |
| Agreed CGK | 18 |
| Nucleotides and nucleosides | 18 |
| Nucleoside analogues | 19 |
| Gemcitabine | 19 |
| Mechanisms of action | 20 |
| Metabolic pathways | 21 |
| Overcoming resistance/intracellular drug delivery issues with nucleoside analogues | 21 |
| Measuring activity | 22 |
| Disputed CGK – issues other than synthetic chemistry | 23 |
| Mechanisms of action and metabolism of NAs in cancer | 23 |
| AraU | 23 |
| dFdUMP | 24 |
| Positional changes | 25 |
| 2' position of the sugar, 5 position of the pyrimidine base | 25 |
| Q position of the sugar | 28 |
| Changing the base | 28 |
| (Bio)isosterism | 29 |
| Prof McGuigan's ProTide work | 31 |
| Potential utility as anti-cancer agents | 32 |
| Disputed CGK – synthetic chemistry | 34 |
| Literature search | 34 |
| Experience of nucleosides | 35 |
| Fluorination conditions | 35 |
| Analytical techniques | 35 |
| The Patents | 35 |
| Teaching of the specification | 35 |
| Quality of the experiments in the Patents | 37 |
| In vitro data | 37 |
| In vivo data | 38 |
| Assessment | 38 |
| Differences in EP365 | 39 |
| Claims in issue | 39 |
| Claim Scope and infringement | 41 |
| Validity | 41 |
| Added matter | 41 |
| The central argument | 42 |
| The law | 43 |
| Domestic law about selection from lists | 47 |
| The EPO case law | 47 |
| T615/95 | 50 |
| T50/97 | 51 |
| T948/02 Great Lakes | 51 |
| T1150/15 Shionogi | 51 |
| G2/10 | 52 |
| Relevance of motive | 53 |
| The preliminary opinion of the TBA on EP190 | 53 |
| The facts | 54 |
| Relevant disclosure of the PCT | 54 |
| Relevant disclosure of EP190 as proposed to be amended | 57 |
| Comparison | 58 |
| Is there added matter? | 58 |
| Alkyl | 59 |
| Conclusion on EP190 added matter | 60 |
| UKIPO comments | 60 |
| EP365 | 60 |
| Industrial applicability and plausibility | 61 |
| Law on capable of industrial application | 61 |
| Law on plausibility | 63 |
| Ab initio plausibility or implausibility | 64 |
| Technical contribution – NuCana's case | 65 |
| Disclosure of the contributions | 67 |
| Cytotoxicity – plausibility facts and assessment | 68 |
| Two aspects of Dr Galmarini's evidence | 69 |
| Assessment | 71 |
| Research tools | 73 |
| Lack of Industrial Application | 74 |
| “Sufficiency”/“existence in fact” | 75 |
| Assessment | 77 |
| Improved intracellular delivery – plausibility facts and assessment | 79 |
| Shepard | 79 |
| Undue burden insufficiency | 80 |
| Terminology | 81 |
| The Law | 81 |
| The standard for undue burden | 82 |
| The role of primary and secondary evidence | 83 |
| Overview of the routes | 84 |
| Two broad categories | 84 |
| Glyceraldehyde routes | 85 |
| Fluorination routes | 85 |
| Fluorination conditions | 86 |
| Fluorination Schemes Document | 87 |
| Choice of successful fluorination conditions used by Concept | 88 |
| Prioritisation | 88 |
| Primary evidence | 89 |
| Prof Micklefield | 89 |
| Prof Davies | 91 |
| Comparison | 91 |
| Secondary evidence: real teams | 92 |
| Idenix | 92 |
| Personnel | 93 |
| Competence of Dr Griffon | 93 |
| Focus on yield | 94 |
| Fluorination conditions | 95 |
| Failure of the glyceraldehyde work | 95 |
| Pharmasset | 96 |
| Mr Clark and Prof Pankiewicz | 96 |
| Overall timing | 96 |
| Glyceraldehyde routes | 97 |
| Fluorination route | 97 |
| Secondary evidence: CRO experiments | 98 |
| Concept | 98 |
| FOB | 99 |
| Analysis | 99 |
| Conclusions | 102 |
INTRODUCTION
In this action the Claimants (together, “Gilead”) seek the revocation of two patents owned by the Defendant, NuCana. They are European Patent (UK) 2 955 190 B1 (“EP190”) and European Patent (UK) 3 904 365 B1 (“EP365”). I will refer to them together as “the Patents”. They are from the same family and although their specifications differ, the differences mostly do not matter and I pick up those points of significance as they arise below. Where I refer to specification paragraph numbers without expressly saying which of the Patents, then I mean EP190.
EP190 and EP365 also differ in their claims, with EP365 being narrower.
The Patents have the same unchallenged priority date of 21 July 2003. As I explain in more detail below, EP190 is under opposition in the EPO with a Technical Board of Appeal (“TBA”) hearing due on 24 March this year. EP365 was only granted on 7 September 2022.
NuCana counterclaims for infringement by Gilead's commercially very successful drug compound sofosbuvir, which is the active ingredient, or one of the active ingredients in its Sovaldi, Harvoni, Vosevi and Epclusa products, which are anti-virals.
NuCana had its own product covered by the claims of the Patents, Acelarin, intended as a cancer drug. It has run into difficulties with clinical trials. That is not of any direct relevance to the issues before me although Gilead sought to emphasise that the Patents teach that the compounds disclosed are cytotoxic, which is desirable in a cancer drug but, Gilead submits, positively to be avoided in an anti-viral.
There is no real challenge to infringement if the claims of the Patents are valid and Gilead took the role of claimant at the trial.
The Patents arise from the work of the late Prof Chris McGuigan of Cardiff University. It will help an understanding of this judgment if I explain at this early stage and at a very high level the technology involved.
For decades it has been known that cancer can be treated with drugs based on nucleoside analogues (also referred to as “NAs”, I will use the full term and the abbreviation interchangeably in this judgment and both are used extensively in the papers in the case). These are compounds which are similar to natural nucleosides but which are modified so that, by one or more mechanisms, they interfere with cells' normal replication and kill them or stop them from growing. This process affects healthy as well as cancerous cells but cancer cells replicate much faster so are hit harder.
A common general knowledge (“CGK”) nucleoside analogue drug which had been successfully used in treating cancer was gemcitabine:
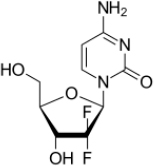
A problem with nucleoside analogues arose with getting them into cells. The reasons for this are relatively complex and are discussed in more detail below. At this introductory level what is significant is that Prof McGuigan's work was concerned with making prodrugs, referred to as ProTides, to try to overcome this problem. The ProTide approach involves phosphoramidate modifications (again, explained below) to nucleoside analogues.
The claims of the Patents are product claims; they are defined by a Markush formula (the permitted substituents vary from claim to claim) and cover ProTide nucleoside analogues as just described in which the nucleoside moiety is gemcitabine or gemcitabine-like. Sofosbuvir falls within the Markush group:
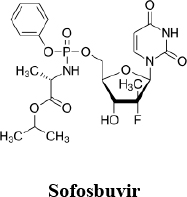
In prosecution and in the opposition proceedings NuCana has successively narrowed its patent claims. At this trial it seeks to amend EP190 in two proposed amended forms (one unconditional and one conditional, the latter being a small extra limitation over the former), and EP365 has claims that are narrower still. NuCana floated a further set of amendments to EP190 in late December 2022, shortly before trial, but abandoned it on the first day of trial.
Gilead attacks the Patents on the following grounds:
i) Added matter: Gilead says that the Markush group definitions of the claims of the Patents are not clearly and unambiguously disclosed in the relevant original application.
ii) Lack of plausibility: Gilead says that the Patents do not plausibly disclose any technical contribution common to substantially all the claimed compounds. NuCana says that there is a plausible technical contribution in relation to the compounds having cytotoxic activity or improved intracellular delivery; it says that the cytotoxicity means that the compounds have the potential to be used in the treatment of cancer. Lack of plausibility is run under the legal heads of:
a) Obviousness over US 2003/0109697 (“Shepard”) on the basis that given the lack of plausibility there is no technical contribution;
b) Insufficiency.
iii) Lack of industrial...
To continue reading
Request your trial
